
Molecular Geometries The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and ⦠Satisfying the octet of the peripheral fluorine atoms will leave 2 electrons, so once again the antimony atom receives a lone pair. The possible electron pair and molecular geometries are: To do so, we first need to draw a Lewis structure for SCl 4. First you have to draw a Lewis structure for the ions, then work out a VSEPR formula, and from that you can determine an appropriate geometry. Step 2: Calculate the total number of valence electrons present. Back to Molecular Geometries & Polarity Tutorial: Molecular Geometry & Polarity Tutorial. This colourless, viscous liquid is a valuable Lewis acid and a component of the superacid fluoroantimonic acid, the strongest known acid, formed when mixing liquid HF with liquid SbF5 in a 2:1 ratio. With two bonding pairs on the central atom and no lone pairs, the molecular geometry of CO 2 is linear (Figure 6.3.3 ). Which direction does the reaction NH3(aq) + H2O(l) = NH4+ (aq) + OH-(aq) shift when solid NH4Cl is added. Identify the elements in these compounds Finally, the molecular orbital theory was proposed to ⦠Lewis dot structures are useful to predict the geometry of a molecule. Now draw the antimony atom in the center surrounded by five fluorine atoms. Basic Rules for Molecules ABn (A = central atom, B = outer atom) Bonding (single and multiple) electron pairs and nonbonding electron pairs (= âlone pairsâ) repel each other and therefore avoid each other, as far as. Antimony pentafluoride is the inorganic compound with the formula SbF5. â is a weakly coordinating anion akin to PF6â. To draw a Lewis structure, start by counting up all the valence electrons available (adding or subtracting as needed to account for the ion's charge). Quiz your students on SbF4- Lewis Dot Structure, bond angle, polar or nonpolar, and molecular geometry using our fun classroom quiz game Quizalize and personalize your teaching. Antimony (Sb) is part of the nitrogen family and so has 5 valence electrons. The ion - has a grand total of 34 valence electrons. , InChI=1/5FH.Sb/h5*1H /q +5/p-5/rF5Sb/c1-6(2,3,4)5, Except where otherwise noted, data are given for materials in their. SO4 2- Molecular Geometry / Shape and Bond Angles - YouTube Get answers by asking now. "Dioxygenyl Salts", National Institute for Occupational Safety and Health, "Chemical Database - Antimony Pentafluoride", National Pollutant Inventory - Antimony and compounds fact sheet, National Pollutant Inventory - Fluoride compounds fact sheet,, Articles with changed ChemSpider identifier, Pages using collapsible list with both background and text-align in titlestyle, Articles containing unverified chemical infoboxes, Srpskohrvatski / ÑÑпÑÐºÐ¾Ñ ÑваÑÑки, Creative Commons Attribution-ShareAlike License, Releases hydrofluoric acid upon contact with water or biological tissues, This page was last edited on 22 January 2021, at 04:30. A quick explanation of the molecular geometry of SBr2 including a description of the SBr2 bond angles. Molecular Geometry: Trigonal Bipyramidal Molecular Shape: T-Shaped Planar. The Lewis structure will show four flourine atoms surrounding a central antimony atom. C almost always forms 4 bonds! Edited by G. The main premise of the VSEPR model is that the electron pairs within the valence shell of an atom repel each other and determine the molecular geometry of the molecule or ion of interest. Does the temperature you boil water in a kettle in affect taste? It is notable for its Lewis acidity and its ability to react with almost all known compounds. But the ion also has a 2- charge, so you have to ADD 2 more valence electrons, bringing the total to 42. 2 Names and Identifiers Expand this section. "A" represents the central atom and n represents the number of bonds with the central atom. The VSEPR notation for these molecules are AX n. , Antimony pentafluoride is prepared by the reaction of antimony pentachloride with anhydrous hydrogen fluoride:, It can also be prepared from antimony trifluoride and fluorine.
How does the hybridization of the sulfur atom change in the series sf2, sf4, sf6? how to#
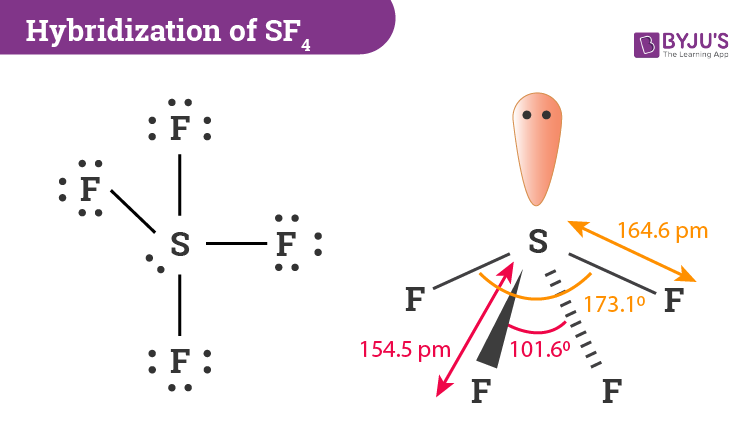
A step-by-step explanation of how to draw the BF4- Lewis Structure. As stated above, molecular geometry and electron-group geometry are the same when there are no lone pairs. 4 bonds / 2 lone pairs / Square planar 4 bonds / O lone pairs / Tetrahedral 4 bonds / 1 lone pair / See saw 5 bonds / O lone pairs / Triganol bipyramidal VSEPR Notation. The geometry of SF 4 is seesaw with an asymmetric electron region distribution around the central atom.
